

Review Article - African Journal of Food Science and Technology ( 2020) Volume 11, Issue 1
Published: 28-Feb-2019, DOI: 10.14303/2141-5455.2020.004
This study attempted the processing and inclusion of insects into a local staple food to endear them to the population. Mature desert locusts (Schistocerca gregaria) and migratory grasshoppers (Acanthacris ruficornis) reared at Egerton University were dried and ground into powder then used to develop composite flours with malted finger millet flour. Proximate analysis and protein digestibility tests of the composite flours were done and their microbial loads (total viable counts, TVC; coliforms; Lactic acid bacteria, LAB; Yeasts and molds) were evaluated. The addition of locust and grasshopper flour increased the protein and fat contents of finger millet flour from 9.20 to between 13.60 and 27.30% and from 1.40 to 3.30–11.40%, respectively. Protein digestibility of the finger millet was improved by malting (from 71.50 to 91.50%) while that of the insect flours was high (approximately 90.00%). The microbial loads in individual products and in composite flours ranged between 106–108 cfu/g for TVC, 105–108 cfu/g for coliforms, 104–107 cfu/g for LAB, and 1.7–2.5 × 107 for yeasts and molds. Our study indicates that locusts and grasshoppers could be incorporated into existing staple foods to generate highly nutritious products that could be recommended to fight malnutrition.
Finger millet, Sustainable protein sources, Insect farming, Malting
The Food and Agriculture Organization (FAO) of the United Nations (UN) has estimated that by the year 2050, the global human population will hit 9 billion (FAO, 2015). Moreover, it is also projected that in the near future, a greater number of the population will enjoy higher standards of living (Makkar, 2018). This will result into greater pressure on the environment, agricultural land, water resources, forests, fish supply and biodiversity, and an increased demand for non-renewable energy (FAO, 2015). All these will increase food demand in particular protein-rich foods; consequently increasing the demand for food of animal origin whose output has to increase by 70% to meet the then demand (FAO, 2013; Floros et al., 2010). With the decrease of the high potential agricultural land suitable for livestock production, there is need for innovative solutions in sustainable animal agriculture. The solutions that have been suggested include reducing the consumption of meat; improving the efficiency along the meat chain; ‘field to fork’, and adopting diets that require less land to produce (van Huis et al., 2015).
The urgency to find alternative sustainable animal protein sources, hunger solutions (low reproduction, farmland) and low-cost farming technology has led to the recognition of insect protein (Adámková et al., 2017). The development of insects such as grasshoppers, locusts, crickets, termites, stink bugs, beetles, caterpillars, flies, and ants as food or feed is an emerging strategy considered to potentially contribute to food security and as a sustainable method for food production (FAO, 2013; Mutungi et al., 2017; van Huis, 2013). Insect farming is advantageous as it leads to lower emission of greenhouse gases and ammonia than conventional livestock and insects have a higher efficiency in converting feed to protein (Gahukar, 2011; Nakagaki and Defoliart, 1991; Oonincx et al., 2010). Insects are rich in high quality protein, polyunsaturated fatty acids, vitamins and minerals (Rumpold and Schlüter, 2013). Insect farming is a low-tech activity and requires low-capital (land and water) investment; hence it can be adopted by low income families (FAO, 2013). Insect production technologies reduce overreliance on conventional feed streams, while bringing valuable ingredients from organic waste materials from agriculture, food industries and other sectors back to the food chain (Rumpold and Schlüter, 2013). In some countries, crickets have been farmed by small-scale farmers for food production (FAO, 2013), and only recently that commercial farming of some insects was adopted (Das et al., 2009; FAO, 2013; Mutungi et al., 2017; Smallstarter, 2014).
Although many insects have been utilized as feed supplements, locusts and grasshoppers stand out since they are edible by humans, are primary consumers and contain large quantities of quality proteins (about 62%), oil (17%) that has essential fatty acids such as linoleic, linolenic acids and oleic acid similar to fish oil, vitamins and minerals (Cheseto et al., 2015; Ramos-Elorduy, 1984). These can thus be tapped to provide inexpensive human food especially for infant and young child feeding in areas where protein and energy are deficient leading to high malnutrition levels. When promoting insects as food or feed, processing them into tasty and attractive forms is one of the major challenges that need to be addressed (van Huis, 2015). In Kenya for example, termites and lake flies were baked, boiled, steamed and processed into crackers, muffins, sausages, and meat loaf and this seemed to encourage entomophagy (Ayieko et al., 2010). Additionally, mixing sorghum and Bambara nuts with caterpillars has been considered to produce a food suitable for children of 10 years and older (Allotey and Mpuchane, 2003). Therefore, integrating the insects into existing foods especially low-protein staple foods such as cereals and tubers could make adoption of the insects much easier.
Finger millet is a cereal that is mainly grown eastern and southern Africa and India used for weaning (Crowley and Carter, 2000). Finger millet is a good source of carbohydrates, calcium, other minerals and fibre. Protein content ranges between 5.60% and 12.70% and among the amino acids, similar to most cereals; lysine is limiting (Singh and Raghuvanshi, 2012). The fat content of finger millet has been reported to range between 1.30 and 1.80%. Finger millet is high (74.40%) in polyunsaturated fatty acids (Antony et al., 1996), with oleic acid being the predominant fatty acid followed by palmitic acid and linoleic acid while linolenic acid is in trace amounts (Sridhar and Lakshminarayana, 1994). Finger millet has a high level of polyphenols that have been found to alleviate some diseases and also reduce the microbial profile of other foods that they are applied to (Viswanath et al., 2009). It has been found that communities that feed on finger millet regularly have low cancer prevalence (Amadou et al., 2011). Enriching finger millet flour with high protein, fat and mineral-rich locusts and grasshoppers to generate composite flours could make it wholesome and useful for weaning and feeding young children. However, maximum utilization of the nutrient potential of finger millet could be limited by the presence of phytates, phenols, tannins and enzyme inhibitors (Singh and Raghuvanshi, 2012). Malting significantly reduces the levels of polyphenols and tannins (by 54%) that are anti-nutritional (Rao, 1994). During the sprouting of the finger millet, there is growth of desirable lactic acid bacteria, which could be beneficial to human health (Singh and Raghuvanshi, 2012).
The aim of the current study was to produce a food product based on locusts and grasshoppers and malted finger millet flour. The effectiveness of these trials are discussed on the basis of the findings related to product development from locusts and grasshoppers and malted finger millet flour, thereby generating composite flours with increased protein and fat contents. Analysis of the products revealed the need for processing insects prior to consumption. Thus, this paper proposes locust/grasshopper-based malted finger millet flour as a new value-added product from reared locusts and grasshoppers.
Sample preparation
The locusts (Schistocerca gregaria) and grasshoppers (Acanthacris ruficornis) that were used were obtained from the insectary at the faculty of Agriculture, Egerton University. They were mature insects of 4 weeks after hatching. The preparation of locust and grasshopper flour was carried out by cleaning of the locusts and freeing them from foreign matter. Inedible parts (appendages) including legs, wings and the heads were then separated before washing of the locusts and oven-drying at 50°C for 48h. The dried locusts were then milled in a laboratory miller to pass a 0.4 mm screen (Babiker et al., 2007) and the flour stored at 9-10°C until further use.
The finger millet that was used for the studies was obtained from the Egerton University farm. The malting of the finger millet was carried out as described by Osuntogun et al. (1989). Briefly, one (1) kg of the grain was washed three times and steeped in 2 L of water for 24 h. Water was changed after every 6 h during steeping. The grains were then washed after steeping and germinated in ventilated cupboards for a period of 1-2 days at an ambient temperature of 28 ± 4°C. Water was sprinkled on the germinating seeds regularly, and the grain was occasionally mixed. After 2 days of germination, the seeds were removed and kilned in an oven at 48 ± 2°C for 24 h. The malted grains were milled to fine flour with a Brabender Quadrumat experimental mill (Siwela et al., 2007) and stored at 9-10°C until further analyses.
Preparation of composite flour
The malted finger millet and the locust flours were mixed to give composite flour (Figure 1). The flours were mixed at predetermined levels so as to obtain a mix that meets the energy and protein requirements of infants. The locust flour substitution took place at 9.6%, 17.3% and 38.4% according to Mbithi et al. (2000).
Proximate Analysis
The moisture content of the finger millet flour, locust flour and composite flour was determined using oven-drying to a constant weight at 105°C (AOAC, 2000). By subtracting the moisture content, the total solid content of the different products and ingredients was estimated. Fat content of the millet, locust and composite samples was determined by Soxhlet extraction using petroleum ether (AOAC, 2000). The protein of the finger millet, locust and composite flours was determined by micro-Kjeldahl procedure using a conversion factor of 6.25 (AOAC, 2000).
The crude protein was calculated as follows; equation (1):
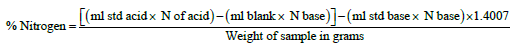
CP=6.25 × % Nitrogen (Where, 6.25 is the conversion factor) Equation (1)
In vitro protein digestibility (IVPD) determination
Two hundred milligrams of sample of locust flour was suspended in 15 mL of 0.1 N HCl with 1.5 mg pepsin in 100 mL conical flask followed by incubation at 37°C for 3 h (AOAC, 2000). The mixture was then neutralized with sodium hydroxide and treated with 4 mg pancreatin (Grade VI porcine) in 0.2 M phosphate buffer containing sodium azide and incubated at 37°C for 24 hrs. Trichloroacetic acid was added to stop the reactions followed by centrifugation at 5000 rpm for 5 minutes. Five milliliters of aliquots were pippeted and analyzed for nitrogen content. The protein digestibility was estimated using eqn. (2).
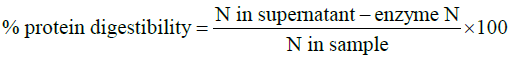 Equation (2)
Equation (2)
Microbial Analysis
Twenty five (25) grams of the test sample was weighed and put into a narrow neck flask containing 225 mL sterile peptone water to obtain 1:10 dilution. Serial dilution of the sample was done up to 10-6 by diluting 1 mL of sample into 10 mL of subsequent dilution bottles. For determination of microorganisms in the samples, pour plate technique was used (Dijk et al., 2007). Total viable Count (TVC) was determined using plate count agar (Techno PharmChem, India) with incubation at 37°C for 48 h. Yeasts and molds were enumerated on potato-dextrose agar (Titan Biotech Ltd, Rajasthan, India) incubated at 28°C for 5 days. Lactic acid bacteria were counted on MRS agar (Becton, Dickinson and Company, USA) and incubation done at 37°C for 48 h. Coliforms were determined by using Violet Red Bile agar (VRBA) (Techno PharmChem, India) and incubation at 37°C for 24 h (Dijk et al., 2007).
Data analysis
Statistical data analysis was performed using the ANOVA procedure of SAS program version 9.3 (SAS Institute, 2011). Statistical significances of differences among treatments were assessed using the Tukey’s Honest Significant Difference (HSD) test.
Proximate composition of flours
The moisture content of finger millet was 7.80 ± 0.20% before it was malted and after malting for two days, the moisture content was estimated to be 9.70 ± 0.30% (Table 1). The average moisture contents of the migratory grasshopper and the desert locust flours were 3.60 ± 0.10% and 8.60 ± 0.20%, respectively, while that of composite flours ranged between 7.70% and 11.70% (Table 1). The data for fat and protein composition of finger millet, migratory grasshopper and desert locust flours is presented in Table 1. The crude protein content of migratory grasshoppers and desert locust flours were 46.00 ± 4.00 and 42.00 ± 2.00%, respectively, while the fat content were 26.00 ± 0.60% and 23.00 ± 0.50%, respectively (Table 1). The protein content of the composite flour increased from 9.20 ± 0.40% for the finger millet to 13.90 ± 0.30% and 13.60 ± 0.30% when the desert locust and migratory grasshopper flours, respectively, were added at 9.6% level. The increment in protein content was proportionate to the amount of insect flour incorporated in the composite flour and the highest protein level (27.30 ± 0.10%) was obtained when desert locust flour was added at 38.4% (Table 1). Similarly, the fat content of the composite flour was increased from 1.40 ± 0.10% of malted finger millet flour to between 3.30 ± 0.24% and 11.40 ± 0.40%, proportionate to the amount of insect flour added (Table 1). Malting the finger millet for two days was found to increase protein digestibility. The protein digestibility of insect and composite flours ranged between 89–91% (Table 1).
| Sample | Moisture Content (%) | Fat Content (%) | Protein Content (%) | Protein Digestibility (%) | Protein Digestibilityc (%) |
|---|---|---|---|---|---|
| Finger millet | 7.80 ± 0.20 | 1.40 ± 0.10 | 9.20 ± 0.40 | 71.50 ± 0.10 | 91.50 ± 0.50 |
| Desert locust | 8.60 ± 0.20 | 23.00 ± 0.50 | 42.00 ± 2.00 | 91.00 ± 0.40 | |
| Migratory grasshopper | 3.60 ± 0.10 | 26.00 ± 0.60 | 46.00 ± 4.00 | 90.00 ± 0.20 | |
| A (9.6%) | 8.10 ± 0.40 | 3.30 ± 0.24 | 13.90 ± 0.30 | 90.10 ± 0.10 | |
| B (9.6%) | 9.80 ± 1.40 | 3.50 ± 0.50 | 13.60 ± 0.30 | 90.00 ± 0.10 | |
| A (17.3%) | 8.00 ± 0.70 | 3.70 ± 0.50 | 16.60 ± 0.60 | 89.80 ± 0.30 | |
| B (17.3%) | 11.00 ± 1.10 | 3.90 ± 0.70 | 17.50 ± 0.40 | 89.50 ± 0.20 | |
| A (38.4%) | 7.70 ± 0.60 | 10.30 ± 0.50 | 27.30 ± 0.10 | 91.10 ± 0.10 | |
| B (38.4%) | 11.70 ± 1.50 | 11.40 ± 0.40 | 26.80 ± 0.50 | 89.40 ± 0.20 |
A, composite flour of malted finger millet mixed with desert locust flour at the rates indicated; B, Composite flour of malted finger millet mixed with migratory grasshopper flour at the rates indicated.
cProtein digestibility after malting finger millet. Value=Mean ± S.D. on dry weight basis. Each value is a mean of 3 replicates.
Microbial contamination
The microbial loads of the desert locusts, grasshoppers, finger millet and composite flours are presented in Table 2. The total viable count of finger millet before malting (7.99 ± 0.18 log cfu/g) was not significantly different (p>0.05) from those of after malting (7.62 ± 0.19 log cfu/g). The migratory grasshoppers had a significantly higher (p< 0.05) TVC (8.19 ± 0.12 log cfu/g) than the desert locusts (7.67 ± 0.14 log cfu/g). The composite flours had a TVC (7.79– 8.01 log cfu/g) that was significantly higher (p> 0.05) than the malted finger millet that was used for mixing. For the coliforms, the loads increased significantly (p> 0.05) during malting. The desert locust had significantly higher (p>0.05) coliform counts (7.97 ± 0.11 log cfu/g) compared to the migratory locusts (5.53 ± 0.55 log cfu/g). The total coliform counts were however not significantly different (p< 0.05) between the composite flours and the malted finger millet and desert locusts from which they were formulated. The LABs reduced significantly (p>0.05) during malting; however the composite flours had similar values to those found in the insects. On the other hand, the yeasts and molds did not vary during malting and were similar in the insect flours and in the composite flours.
| Sample | TVC | Coliforms | LAB | Yeasts and molds |
|---|---|---|---|---|
| Finger millet (0 day) | 7.99 ± 0.18A,B | 5.72 ± 0.88B | 7.28 ± 0.37A | 7.43 ± 0.20A |
| Finger millet (2 days) | 7.62 ± 0.19B | 7.35 ± 0.33A | 5.01 ± 1.08B | 7.66 ± 0.23A |
| Desert locusts | 7.67 ± 0.14 B | 7.97 ± 0.11 A | 7.88 ± 0.20 A | 7.82 ± 0.15A |
| Migratory grasshopper | 8.19 ± 0.12 A | 5.53 ± 0.55 B | 4.88 ± 0.65 B | 7.47 ± 0.19 A |
| A (9.60%) | 7.79 ± 0.20A | 6.66 ± 0.93A | 7.56 ± 0.33A | 7.81 ± 0.27A |
| A (17.30%) | 7.98 ± 0.17A | 6.70 ± 0.31A | 5.76 ± 0.84B | 7.66 ± 0.17A |
| A (38.40%) | 8.01 ± 0.17 A | 6.90 ± 0.40A | 5.82 ± 0.83B | 7.48 ± 0.19A |
In this study, desert locusts and grasshoppers were ground into powder after drying. They were then used as ingredients to make enriched finger millet porridge flour (LocusWean). The moisture content of the insects and composite flours ranged between 3.60 ± 0.10% and 11.7 ± 1.50%, which compares to dehydrated products such as spices, nuts, seeds and flour (Schmidt and Fontana, 2008). At these moisture contents, the growth of most microorganisms is limited and the products can have long shelf lives (Fasolato et al., 2018). The crude protein of the insects ranged between 42.00 ± 2.00% and 46.00 ± 4.00 based on the dry weight. This range was similar to those recorded by Ramos-Elorduy et al. (1997), which ranged between 40% and 60% crude protein. The composite flours had improved protein content compared to the finger millet flour. This proved that our reared desert locusts and grasshoppers can be a good protein supplement. On the fat content, the insects had fat content ranging between 23 and 26%, which is lower than 41-43% that were recorded for edible grasshoppers (Ruspolia nitidula), but higher than that of other reported grasshopper species such as small (6.1%) and large grasshoppers (3.3%) and other edible insects in western Nigeria including a grasshopper species, Zonocerus variegatus (3.8%) (Ssepuuya et al., 2017). These differences could be attributed with species diversity, growing conditions, and the stage of harvesting. However, the fat contents of the insects reported in this study were higher than that reported for meat, pork, and fish, all of which average less than 22% (Ssepuuya et al., 2017). The fat content in the composite flours was improved significantly to between 3.3 ± 0.24% and 11.4 ± 0.40%. Incorporation of desert locust/ grasshopper into finger millet flour is of particular interest because the fat from the insects has been reported to contain essential fatty acids such as linoleic, linolenic acids and oleic acid similar to fish oil (Ssepuuya et al., 2017). The enrichment of finger millet with the insects increased the protein and fat contents in the composite flours that makes them potential candidates for weaning babies compared to finger millet alone since the protein content of 15–20%, fat content of around 10%, carbohydrate content of 60–70% and total ash content of less than 5% are desired for weaning foods (Kumkum et al. 2013).
Protein digestibility of finger millet was improved with malting as similarly reported by Arora and Khetarpaul (2011). Malting converts polysaccharides and oligosaccharides into simple sugars while proteins are broken into amino acids and peptides, improving their digestibility, hence improved nutritive value. Malting also increases the moisture content for the seeds to germinate that influences the content of antioxidants e.g., phenols, flavonoids, tannins, phenolic acids compounds and vitamins E and β-carotene (Singh and Raghuvanshi, 2012). Malting reduces the content of some anti-nutritional components such as phytic acid (Krishnan et al., 2012). During malting, functional bioactive components including antioxidants and vitamins are also generated (Hejazi and Orsat, 2016). Therefore the combination of malted grains and protein and fat rich insects could generate nutritive and inexpensive products such as infant formula, complementary food products, and composite flours or food blends suitable for poor people who have higher cases of malnutrition. The protein digestibility of the composite flours compared to that of beef (0.92) (Hoffman and Falvo, 2005).
The TVC and yeasts and molds levels of 107-108 cfu/g were found in all the products. Although particular microbiological criteria for insects utilized as human food have not been developed, the values for process hygiene criteria for minced meat spelled in the European Union Regulation (EC) No. 1441/2007 can be applicable for insects (SHC and FASFC, 2014). The TVC of the minced meat as per this guideline is 5.7–6.7 log cfu/g while that of yeasts and molds is <4.0 log cfu/g. In this study, the numbers obtained were higher than these values though they were consistent with other values in the studies of mealworms and crickets (Klunder et al., 2012; SHC and FASFC, 2014). Coliforms and lactic acid bacteria were also detected in the products in large numbers. The presence of coliforms, the high TVC, and yeasts and molds levels in both insects and flours indicates unhygienic handling or for the insects, they could have resulted from the release of microbiota from the insect’s intestines of the insects during milling then distributed throughout the product, from the raw materials and through contacting the handling equipment (Mmari et al., 2017). Moreover, insects are rich in nutrients and could provide a favorable environment for microbial multiplication. The drying conditions that were used could not fully kill the microbiota from the insects’ intestines; therefore applying additional processing procedures such as blanching prior to milling could reduce the number of organisms (Fasolato et al., 2018; Klunder et al., 2012; Megido et al., 2017). The lactic acid bacteria (LAB) in the insect flour could have originated from the insect gut or on the skin of the insects. On the other hand, thermophilic LAB could not be inactivated during oven drying. These could thus spoil products inadvertently subjected to favorable conditions during storage (Klunder et al., 2012). For the millet flour, the microorganisms from the environment or on the finger millet could have multiplied during malting. Malting increases the level of total solids in finger millet in particular, monosaccharides increases the ability of these microorganisms to multiply especially the coliforms (Adebiyi et al., 2018). These results indicate that additional processes such as fermentation could be applied to the composite flours to reduce the number of contaminants such as coliforms through the action of increased acidity (Klunder et al., 2012).
In conclusion, desert locusts and grasshoppers were processed and mixed with malted finger millet to make enriched composite flours. The addition of insect flour to the finger millet flour increased the protein and fat content and possibly other nutrients to levels that make the composite flours suitable candidates for weaning or be consumed by broad categories of the population. Malting of the finger millet improved its protein digestibility. This demonstrated that these methods could be employed to generate highly nutritious foods that could be useful in fighting malnutrion especially in sub-Saharan Africa where malnutrition is highly prevalent. However, hygienic handling, standardization of the malting process and additional processing steps are recommended so that products free of/ low in microbial contamination are produced. It will be of interest also to extract nutrients such as proteins and fats from the insects for the development of a diversity of products.
This research received funding (RC102194 Egerton) from the Global Center for Food Systems Innovation (GCFSI) of Michigan State University, which is sponsored by the United States Agency for International Development (USAID).
The authors acknowledge the support given by the Division of Research and Extension and the Centre of Excellence in Sustainable Agriculture and Agribusiness Management (CESAAM) of Egerton University, Kenya during the conduct of the research.
The authors do not have any conflict of interest to declare