

Case Study - African Journal of Food Science and Technology ( 2023) Volume 14, Issue 1
Received: 10-Dec-2022, Manuscript No. AJFST-22-83059; Editor assigned: 15-Dec-2022, Pre QC No. AJFST-22-83059; Reviewed: 19-Dec-2022, QC No. AJFST-22-83059; Revised: 30-Nov-2022, Manuscript No. AJFST-22-83059; Published: 12-Jan-2023, DOI: 10.14303//ajfst.2023.002
Chronic toxicity could occur in humans when “gaari” with excess permissible amount of cyanide is consumed which negates food security. This study assessed the levels of cyanide in the “gaari” sold in Sango, Apete, and Bodija markets in Ibadan, Oyo State, Nigeria. Three most popular, locally processed commercial “gaari” named: Oyo (A), Ilora (B) and Egba (C) were obtained from each of the three markets and were pooled for residual cyanide content analysis using titrimetric and picrate test method. pH, swelling capacity, and bulk density were determined using standard methods. The cyanide content level ranged between 2.51 ± 0.003 to 3.72 ± 0.05mg/ kgHCN in Sango market, 2.51 ± 0.003 to 3.67 ± 0.05mgHCN/kg in Apete market and 2.53 ± 0.003 to 3.78 ± 0.05mgHCN/kg in Bodija market. Egba “gaari” had the highest value of cyanide content in the three markets. The mean cyanide content of the three “gaari” at Sango, Apete and Bodija market were 3.20, 3.15 and 3.21mgHCN/kg respectively. The picrate test ranged from 2-3 giving a yellow-yellow-orange coloration. Their pH values ranged from 4.15 to 5.47, while the swelling capacity was 2.87% - 3.57%. The mean bulk density of “gaari” samples A, B, and C were 0.535g/cm3, 0.542g/cm3, 0.503g/cm3 respectively. The cyanide content of the locally processed “gaari” were below the tolerable standard limit of 5-10mg/kgHCN and as such safe for human consumption. Considering food security and health risk to consumers, it is important to assess other cassava products in Ibadan and beyond for their cyanide levels.
Cyanide, Fermentation, Gaari, Permissible limit, Food security.
Cassava (Manihot esculenta crantz) is a staple meal in Nigeria that is growing in popularity as the country's population grows (Oluwole et al., 2004). Nigerian cassava production is by far the largest in the world; a third more than production in Brazil and almost double the production of Indonesia and Thailand (FAO, 2007). Fresh tubers are highly perishable and cannot be stored or kept fresh for more than a few days after harvest without any degradation in quality (Uchechukwu-Agua et al., 2015). Cassava, is processed into dried products in many ways in different parts of the world to satisfy local demands, tastes, and traditions for use and storage such as chips, cassava flour, and fufu (akpu) (Balogun et al., 2012; Oyeyinka et al., 2019b). The most common form of cassava consumed in West Africa is a roasted granular product made from peeled, grated, and fermented cassava tubers known as “gaari” (Sanni et al., 2008). Gaari is a product obtained by fermenting (approximately 72 hours), draining and roasting peeled, washed and grated fresh cassava roots. In Nigeria, up to 70% of the cassava roots harvested are processed into gaari. Gaari is usually flavored with water and sugar and eaten as a refreshing snack drink, or poured into hot water in sufficient quantity to form a firm pudding (Eba) (Laya et al., 2018) to accompany soups and stews. It is typically processed using manual and mechanical processing methods. There is a possibility that chronic toxicity can result from the ingestion of “gaari” that contains excess of the permissible amount of cyanide (Adindu et al., 2003).
Hydrocyanic acid (HCN), sometimes known as cyanide, is a toxic chemical compound found in high concentrations in raw cassava root. However, the presence of cyanide constitutes a major limitation to the utilization of cassava as human food (Asegbeloyin & Onyimonyi, 2007). HCN is produced by hydrolysing the two cyanogenic glucosides found in cassava tuber; linamarin and lotaustralin (Bradbury & Denton, 2010). Raw cassava can contain 130-200mg HCN/ kg (Mburu et al., 2012). When plant tissues are injured during harvesting or processing, the natural enzyme linamarase can hydrolyse the cyanogenic glucosides to hydrocyanic acid (Marcus & Adesina, 2001). According to Orjiekwe et al. (2013) there are several health disorders which have been associated with regular intake of sub-lethal quantities, some of which have resulted into outright poisoning and death due to cyanide intake from consumption of poorly processed cassava products. The authors further stated that health risks caused by ingestion of these chemicals include the exacerbation of goiter, cretinism and cardiovascular diseases such as encephalopathy and neuropathy, while severe cyanide poisoning can lead to heart, brain and optic nerve degeneration. As a result, some processing procedures have been developed to minimize their toxicity. The fermentation procedure has been shown to improve cassava detoxification by releasing hydrocyanic acid. Apart from detoxification, this process also helps in flavour development and the overall improvement of the quality of the final product. Sun drying, soaking, and fermentation, followed by drying or roasting, are examples of these processes (Irtwange & Achimba, 2009; Laya et al., 2018, Nambisan, 2011). It is thereby important to assess cassava products because of some occasional reports of death, several abdominal upsets and other discomforts following it consumption that have been reported. This is due to the fact that there is increasing demand for “gaari” and this makes some commercial centers to process in a hurry without allowing for sufficient fermentation to cause appreciable cyanide reduction. Therefore, this study aimed to assess the levels of cyanide in the most popular “gaari” being sold in major markets (Ekpechi, 1967).
Collection of samples
Three locally processed commercial “gaari” Oyo (A), Ilora (B) and Egba (C) were obtained from three selected Ibadan markets (Sango, Apete and Bodija markets) in Oyo State, Nigeria. 200g of each sample was labelled in tightly sealed containers and kept in field cellophane bags prior to analysis.
Processing and analysis of samples
Cyanide content of gaari samples were measured by alkaline titration method according to method described by (AOAC, 2017). Approximately 10 g of the sample was mixed with approximately 100 ml distilled water in a distillation flask; the mixture was distilled and approximately 200 ml of the distillate collected in a volumetric flask containing 25 ml of 2.5% NaOH solution. A portion of 8 ml of 6.0 NH4 OH and 2.0 mL of 5% KI indicator solution was then added to 100 ml of distillate and titrated against 0.02N silver nitrate (AgNO3) solution. The end point was indicated by a faint permanent turbidity appearance. The HCN content was calculated as: 1 ml of 0.02 N Silver Nitrate being equivalent to 1.08 mg of HCN per 10 g and then expressed as HCN mg/kg of sample (Nambisian, 2011).
Picrate analysis
This is a qualitative determination of the cyanide potential of vegetative materials. It was estimated using the method of (Bradbury et al.,1999). The colour of the picrate papers was matched against the shades of colour of the colour chart supplied with 1 and 9 representing the lightest and deepest colour, respectively, which serves as the index of the cyanide potential of the gaari sample.
Swelling capacity
The swelling capacity was determined according to the method of (Sanni et al., 2001). A 50ml glass measuring cylinder was filled with “gaari” samples to the 10ml mark. Distilled water was added at room temperature (25+-30oC) to give a total volume of 50ml, the top of the cylinder was tightly covered and the contents mixed by inverting the cylinder, after 2mins, the cylinder was inverted again and then left to stand for 3mins (5mins total time) and the final volume occupied by the “gaari” was recorded. The swelling capacity was thus determined by dividing the volume of the “gaari” in water by the initial volume of “gaari”.
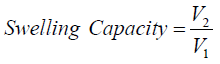
V1=Initial volume of “gaari”, V2 = final volume
Bulk density
Bulk densities were determined in triplicate in a weighed 250ml cylinder according to Picker-Freyer & Brink (2006) with slight modification. 100g of the sample was gently filled into the cylinder. Bulk volume was read and bulk density calculated. The cylinder was tapped at least 50times to a constant volume. Tapped volume was read and tapped density calculated.
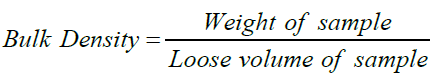
Determination of pH content of gaari samples
The pH of the samples was determined following the method described by Ogiehor & Ikenebomeh (2006). 10g of each sample were homogenized in 100 ml of distilled water and the pH of the suspension determined using a reference glass electrode pH meter (Mettler-Toledo GmbH Switzerland, SevenCompact S210).
Data analysis
Descriptive statistics was done on the data using Microsoft Excel Workbook 2013. The results’ data were analysed using Microsoft Office workbook 2013 by finding mean and standard deviation. Histograms charts were used to plot the data where applicable to identify patterns of relationship within them.
In the study, the mean cyanide content level of the samples Oyo (A), Ilora (B) and Egba (C) from the three markets were 2.51 ± 0.003, 3.32 ± 0.05, and 3.72 ± 0.05 mg/kgHCN respectively as shown in Figure 1. At Sango market, as shown in Table 1, the content value ranges from 2.51 ± 0.003 to 3.72 ± 0.05mg/kgHCN showing Egba gaari with the highest value. The cyanide content at Apete market ranged from 2.51 ± 0.003 to 3.67 ± 0.05mgHCN/kg as shown in Table 1. At Bodija market, the level ranged from 2.53 ± 0.003 to 3.78 ± 0.05mgHCN/kg as shown in Table 1. At the three markets, Egba gaari had the highest cyanide content with little difference compared to Oyo and Ilora “gaari” as shown in Figure 1. Therefore, the mean cyanide content of the three “gaari” popularly being sold at each market were 3.20, 3.15 and 3.21mgHCN/kg and these are safe for consumption regardless of the market location as shown in Figure 2.
| Gaari samples | Market | ||
|---|---|---|---|
| Sango (mg/kgHCN) | Apete (mg/kgHCN) | Bodija (mg/kgHCN) | |
| Oyo (A) | 2.51 ± 0.003 | 2.51 ± 0.003 | 2.53 ± 0.003 |
| Ilora (B) | 3.38 ± 0.05 | 3.27 ± 0.05 | 3.31 ± 0.05 |
| Egba (C) | 3.72 ± 0.05 | 3.67 ± 0.05 | 3.78 ± 0.05 |
Picrate test result
The colour score ranged from 2-3 out of 9 as shown in Table 2, giving a yellow-yellow-orange coloration. Oyo “gaari” had a yellow colour which showed a very low cyanide content compared to Ilora and Egba with yellow-orange colour.
| Gaari samples | Market | ||
|---|---|---|---|
| Sango | Apete | Bodija | |
| Oyo (A) | 2 | 2 | 2 |
| Ilora (B) | 3 | 3 | 3 |
| Egba (C) | 3 | 3 | 3 |
Bulk density
The mean bulk density reading of each “gaari” samples A, B, and C were 0.535g/cm3, 0.542g/cm3, 0.503g/cm3 respectively as shown in Figure 3. Ilora sample had the highest bulk density in all the three markets. This was followed by Oyo, while Egba samples had the least bulk density as shown in Figure 4.
Swelling capacity of the samples from the markets
The average swelling capacity reading from the three markets for the samples Oyo “gaari”, Ilora “gaari” and Egba “gaari” were 3.00%, 2.87% and 3.57% respectively as shown in figure 5. Therefore, Egba (C) “gaari” showing a high swelling capacity, followed by Oyo “gaari” compared to Ilora “gaari” as shown in Figure.5 & Figure 6.
pH readings of the samples
The pH values ranged from 4.15 to 5.47 as shown in Table 3. The Egba sample showing low pH due to it high cyanide content level compared to Oyo and Ilora “gaari” with low cyanide level as shown in Figure 7.
| “Gaari” samples |
Market | ||
|---|---|---|---|
| Sango | Apete | Bodija | |
| Oyo (A) | 4.8 | 5.1 | 5.47 |
| ILORA (B) | 4.6 | 4.5 | 4.41 |
| EGBA (C) | 4.3 | 4.4 | 4.15 |
Gaari is widely recognized as a cassava product. In the study, the average cyanide content level of the “gaari” samples popularly being sold in the three markets were below the cyanide content tolerable standard limit of 5-10mg/kgHCN. The implication here is that local cassava processors in the producing community are already using best practices to process their cassava products and that local consumers are not at risk when consuming the products. cassava. It is widely acknowledged that lengthy fermentation typically 2-3 days dramatically reduces the amount of cyanide and acts as a key detoxification phase (Oyeyinka et al., 2019b; Nambisan, 2011; Lambri et al., 2013). Also, sun drying and proper frying of “gaari” are generally believed to lead to substantial decrease in residual cyanide. The mean cyanide content of the three “gaari” at Sango, Apete and Bodija market were 3.20, 3.15 and 3.21mgHCN/kg respectively which all “gaari” are safe for consumption as compared to other similar studies. Those studies reviewed “gaari” being sold at the market whether it is safe for consumption by the consumers. It is necessary to apply proper processing procedures so as not to have toxic effect on consumers because this is due to the fact that there is increasing demand for “gaari” and this makes some commercial centers to process in a hurry without allowing for sufficient fermentation to cause appreciable cyanide reduction. Although the cyanide levels in the test samples used in this study are within the FAO's acceptable level, while epidemiological studies have shown that exposure to small doses given over a long period of time can result in increased blood cyanide levels with the following symptoms: dizziness, headache, nausea and vomiting, rapid breathing, restlessness, weakness, and even in severe cases paralysis, nerve lesions, hypothyroidism, and miscarriage (Soto-Blanko et al., 2002; Rachinger et al., 2002, Montagnac et al., 2009). According to Orjiekwe et al. (2013), who reported that acute toxicity in humans causes death while chronic toxicity has been associated with goiter and tropical ataxic neuropathy. The government's relevant authorities should therefore make a concerted effort to educate these rural cassava processors on contemporary cassava processing procedures and the health risks posed to consumers by using short-cut processing techniques. To avoid sharp practices, millers should conduct regular routine monitoring of cyanide levels in cassava products (Onwuka, 2005).
The present study revealed that the “gaari” samples being sold at these Ibadan major markets are safe for consumption. The cyanide levels in the “gaari” samples screened contained tolerable levels of cyanide which is relatively very safe and within the acceptable limit of 5-10mgHCN recommended by FAO and WHO according to. It has been evidently shown that these samples were found to have been fermented for 2-3 days prior to frying which is enough for its detoxification. Consider the fermentation process to be an important detoxification step in “gaari” production. It is therefore necessary to stop fermenting cassava for less than two days and it should never be eaten raw, no matter how small the cyanide content is. Extension workers are to be informed, so to educate the communities that enjoy the practice of fermenting cassava in less than two days in order to prevent chronic toxicity in light of the health risk posed by cumulative residual cyanide consumption from cassava. Considering the food security and health risk of consumers, it is important to assess other products of cassava in Ibadan and beyond for the cyanide level to ascertain how nutritionally secure they are.
Adindu MN, Olayemi FF, Nze-Dike OU (2003). Cyanogenic potential of some cassava products in Port Harcourt markets in Nigeria. J Food Compos Anal. 16: 21-4.
Indexed at, Google Scholar, Cross Ref
Asegbeloyin JN, Onyimonyi AE (2007). Effect of different processing methods on the residual cyanide of Gaari. Pak J Nut. 6: 163-166.
Association of Official Analytical Chemists (AOAC) (2017). Official methods of analysis. 17th edition. In: Association of official Analytical Chemists, Rockville.
Balogun M, Karim O, Kolawole F, Solarin A. (2012). Quality attributes of tapioca meal fortified with defatted soy flour. Agrosearch, 12: 61-68.
Indexed at, Google Scholar, Cross Ref
Bradbury JH, Denton IC (2010). Simple method to reduce cyanogens content of garri made from cassava. Food Chem Toxicol. 123: 840-845.
Bradbury MG, Egan SV. & Bradbury JH (1999). Determination of all forms of cyanogens in cassava roots and cassava products using picrate paper kits. JSFA. 79: 593-60I.
Ekpechi OL (1967). Pathogenesis of endemic goiter in Eastern Nigeria. Brit J Nutr. 21: 537-545.
Indexed at, Google Scholar, Cross Ref
FAO (2007). Cassava production statistics. Rome, Italy.
Irtwange S, Achimba O. (2009). Effect of the duration of fermentation on the quality of gaari. Curr Res J Biol, 1: 150-154.
Lambri M, Fumi MD, Roda A, De Faveri, DM (2013). Improved processing methods to reduce the total cyanide content of cassava roots from Burundi.AJB.12: 2685-691.
Laya A, Koubala BB, Kouninki H, Nchiwan Nukenine E (2018). Effect of harvest period on the proximate composition and functional and sensory properties of gaari produced from local and improved Cassava (Manihot esculenta) varieties. Int J Food Sci. 6241035, 1-15.
Indexed at, Google Scholar, Cross Ref
Marcus AA, & Adesina BS (2001). The Effect of Cooking Time on Root Mealiness and Taste of Cassava Tuber. NJFT. 1: 113.
Mburu FW, Swaleh S, Njue W (2012). Potential toxic levels of cyanide in cassava (Manihot esculenta Crantz) grown in Kenya. Afr J Food Sci. 6: 416-420.
Indexed at, Google Scholar, Cross Ref
Montagnac JA, Davis CR, Tanumihardjo SA (2009). Processing techniques to reduce toxicity and antinutrients of cassava for use as a staple food. Compr Rev Food Sci Food Saf. 8: 17-27.
Indexed at, Google Scholar, Cross Ref
Nambisian B (2011). Strategies for elimination of cyanogen from cassava for reducing toxicity and improving food safety. Food Chem Toxicol. 49: 690-693.
Indexed at, Google Scholar, Cross Ref
Ogiehor IS, Ikenebomeh MJ (2006). The effects of different packaging materials on the shelf stability of garri. AJB. 5: 2412-2416.
Oluwole OB, Olatunji OO, & Odunfa SA (2004). A Process technology for conversion of dried cassava chips into garri. Niger Food J. 22: 65-73.
Onwuka GL (2005). Food Analysis and Instrumentation. Theory and Practice. Lagos, Nigeria pp122-128: Naphthal print, Surulere.
Orjiekwe CL, Solola A, Iyen E, Imade S (2013). Determination of cyanogenic glycosides in cassava products sold in Okada, Edo State, Nigeria. Afr J Food Sci. 7: 468-472.
Oyeyinka SA, Ajayi OI, Gbadebo CT, Kayode RM, Karim OR et al. (2019b). Physicochemical properties of gaari prepared from frozen cassava roots. LWT Food Sci Technol. 99: 594-599.
Indexed at, Google Scholar, Cross Ref
Picker-Freyer KM, Brink D (2006). Evaluation of Powder and Tableting Properties of Chitosan. AAPS Pharm Sci Technol. 7: 3.
Indexed at, Google Scholar, Cross Ref
Rachinger J, Fellner FA, Stieglbauer K, Trankler J (2002). MR changes after acute cyanide intoxication. Am J Neuroradiol. 23: 1398-1401.
Sanni LO, Ikumola DO, Sanni SA (2001). Effect of length of fermentation and varieties on the qualities of sweet potato gaari. 208- 211.
Sanni L, Adebowale A, Awoyale W, Fetuga G (2008). Quality of gaari (roasted cassava mash)in Lagos State, Nigeria. Niger Food J. 26: 125-134.
Soto-Blanco B, Maiorka PC, Gorniak SL (2002). Neuropathologic study of long term cyanide administration to goats. FCT. 40: 1693–1698.
Indexed at, Google Scholar, Cross Ref
Uchechukwu-Agua AD, Caleb O., & Opara UL (2015). Postharvest handling and storage of fresh cassava root and products: a review. Food Bioproc Tech. 8: 729-748.
Indexed at, Google Scholar, Cross Ref
Citation: Fisayo O.O & Onoyinka A.A. (2023). Cyanide content and physical properties of commercial “gaari” samples from different markets in ibadan, oyo state, nigeria. AJFST. 14: 002